Epilepsy Genetics
Epilepsy is a clinically heterogeneous disease with diverse aetiologies. Advances in molecular genetics over the last ten years have led to an explosion of novel genes implicated in monogenic and complex genetic epilepsies. Therefore, genetic testing now has become a critical part of the diagnostic evaluation of adults and children with epilepsy to identify genetic epilepsy syndromes, guide treatment, optimize genetic counseling, and bring closure and peace of mind to the families of those with a genetic disease whether treatable or not.
The Ontario Epilepsy Genetic Testing Program (OEGTP)
On October 1, 2020, the Ontario Ministry Health [MOH] established epilepsy gene panel testing in Ontario as a publicly funded service for patient's who meet the ministry's eligibility criteria. Therefore, after this date, the MOH's Out-of-Country/Out-of-Province (OOC/OOP) Prior Approval Program for Laboratory Testing will no longer be accepting applications for epilepsy panel testing and ordering physicians will be redirected to the testing provided in Ontario.
Specifically, based on the recommendations of an MOH epilepsy genetics advisory group, the Ontario Epilepsy Genetics Testing Program [OEGTP] provides evidence-based epilepsy gene panels (PMID:31587668) for properly selected epilepsy patients. The latest revision of panel genes was performed in December 2023. This genetic testing is carried out at the Division of Molecular Diagnostics in the Pathology and Laboratory Medicine Department at London Health Sciences Centre.
Access to epilepsy genetic testing now is available through a new requisition (updated in December 2023) that is designed to provide clinical eligibility criteria and evidence-based gene panel selection OEGTP Requisition.
Project Echo Epilepsy Across the Life Span and the OEGTP
With the advent of OEGTP Epileptologists and Neurologists affiliated with Ontario District and Regional Epilepsy Centres, FRCP Neurologists with a minimum of six months of training in epilepsy and EEG, and Geneticists can order genetic testing in epilepsy patients. In addition, Ontario licensed physicians who acknowledge completion of the Continuing Medical Education (CME) certified epilepsy genetics curriculum in Project ECHO Epilepsy Across the Life Span will be able to order genetic testing for their epilepsy patients, where indicated, through the OEGTP. Project ECHO Epilepsy Across the Lifespan is a provider education opportunity targeted at Family Physicians, Paediatricians and Community Neurologists.
Project ECHO (Extension for Community Healthcare Outcomes) is an innovative model for medical education that uses video-conferencing with case-based learning to connect specialist multidisciplinary teams at academic health centres to community healthcare providers. In this way Project ECHO: Epilepsy Across the Lifespan delivers pediatric and adult epilepsy curricula to improve the quality and accessibility of epilepsy care in Ontario. ECHO ONTARIO The genetics of epilepsy now is an integral part of the Project ECHO Epilepsy Across the Life Span curricula.
This one-credit-per-hour Group Learning program meets the certification criteria of the College of Family Physicians of Canada and has been certified by Continuing Professional Development, Faculty of Medicine, University of Toronto for Mainpro+ credits. These sessions are also an Accredited Group Learning Activity (Section 1) as defined by the Maintenance of Certification Program of the Royal College of Physicians and Surgeons of Canada, approved by Continuing Professional Development, Faculty of Medicine, University of Toronto.
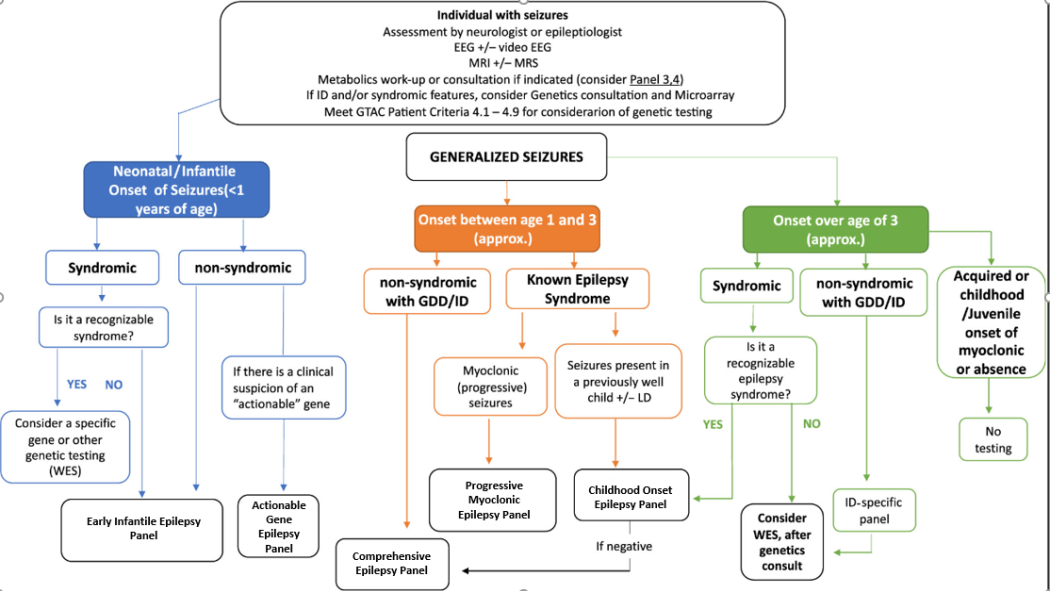
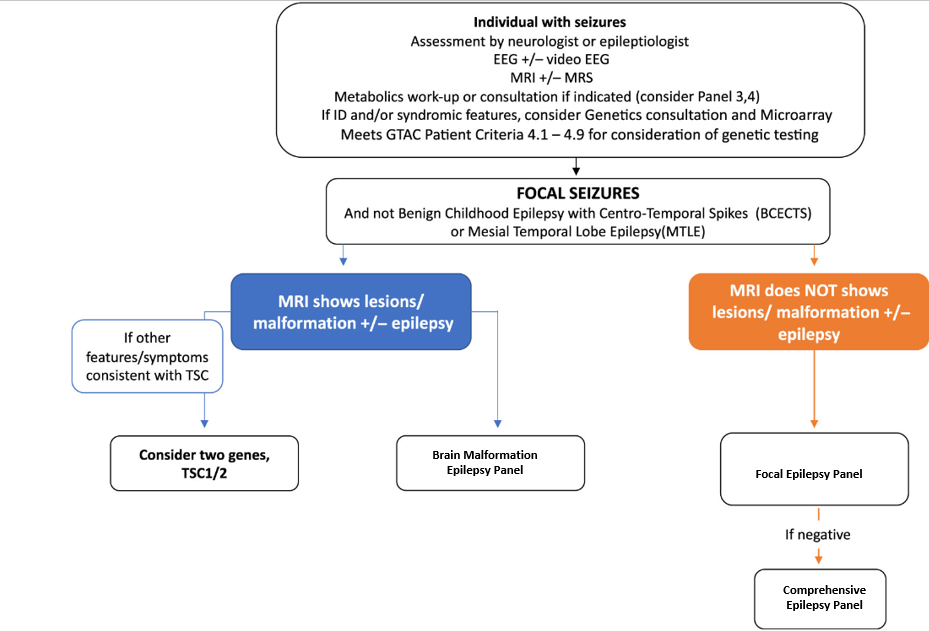
Test Indications
This Epilepsy panel test is a deep sequencing NGS assay designed as a rule out sequencing and copy number analysis test for all coding sequences of all genes tested. Content is designed by a panel of clinical experts Ontario MOHLTC Genetic Epilepsy Working Group to include majority of genes associated with epilepsy as the cardinal clinical presentation. In patients where epilepsy is not the cardinal clinical feature, and genetic etiology is suspected, other genetic and genomic analyses and clinical genetics referral may be considered.