Ensure that patient and health care provider safety standards are met during this procedure including:
| |
PROCEDURE | |
Ordering Blood Cultures in CCTC: Any order for blood cultures should consist of a MINIMUM of 2 complete sets of cultures. Each set of cultures should be drawn from a different draw (e.g., 2 different venipuncture sites or a venipuncture plus line site(s). RNs carry a medical directive to order cultures when indicated. Line details should include the type of intravascular device (e.g,. triple lumen, arterial line, PICC, HD catheter), the site (e.g., right IJ) and the date of insertion. Be sure to specify the type of central line. Line details must be entered at the time the orders are placed. The purpose of drawing 2 or more cultures is to increase the sampling yield and to help to rule out contamination. In the rare event that cultures can only be obtained from a single draw or site, the reason for the single draw should be documented. Do not draw two sets of cultures from the same draw (as this would still be a single draw specimen and lead to misinterpretation of results). Nurses are accountable to ensure that cultures are drawn as per protocol. If a physician orders a single blood culture, the nurse is authorized to obtain a second set. Ideally, blood cultures for patients with indwelling intravascular lines should include one venipuncture plus any indwelling line cultures. In CCTC, all blood culture orders should be entered by a nurse to facilitate the entry of details about the line insertion. This information cannot be entered once the order has been placed. If a physician enters an order for blood cultures (and this information has not been entered with the order), cancel the blood culture order should be cancelled and re-entered with the necessary information. This should include the date of insertion, the location of insertion (e.g., R IJ) and type of catheter (e.g., specify whether the central line is a PICC or dialysis line). Quick orders for blood cultures are for "nurse to order cultures" or "nurse to pan culture". | |
. | Indications for Blood Cultures
Notes: Many patients are admitted with sepsis as a differential diagnosis; prompt cultures are indicated. This should Ideally occur prior to antimicrobial therapy initiation but should not delay treatment. Staph aureus in the blood is associated with endocarditis. Staph aureus and yeast can be difficult to eradicate. Line removal is generally required (including tunneled catheters). Blood cultures are typically repeated until they become negative. The time when the cultures are first negative helps to determine the duration of antimicrobial therapy. |
| 2. | Identify Number of Samples to be Collected See Blood Culture Ordering Decision Tree: 1. If the patient has no intravascular lines, draw 2 sets of peripheral cultures from 2 different draws (different lines or puncture sites).
2. If the patient has intravascular lines in place that are > 24-48 hours, draw cultures and request a "CAB" assessment as follows:
3. If the patient has a previously established line that is being removed and obtain cultures from the line and at least one other site and send the tip for culture (done semiquantitatively). NOTE: A Catheter Associated Bacteremia (CAB) assessment will only be performed if a venipuncture sample is included (and labeled as a venipuncture sample). It required lab notification for appropriate setup of cultures. If any indwelling line becomes positive more than 2 hours before the venipuncture culture first became positive, the blood stream infection (bacteremia) is unidentified as a CATHETER ASSOCIATED BLOOD STREAM INFECTION. If all blood cultures become positive within a 2 hour window, the infection is not considered to be catheter associated. All samples must go to the lab at the same time so that they can be setup together. A newly established line can be considered a "peripheral stab" ONLY if it is newly established and has not been previously used for blood drawing. If the sample is drawn at the time of insertion, identify this as a "peripheral culture" in the lab orders. If a peripheral culture cannot be obtained, report this under "Comments" in Power Chart (see item 7) and document in the AI flow sheet. (A CAB assessment will not be performed without a sample that is labeled as "venipuncture"). Simultaneous results from multiple sites aids in the interpretation of the results (e.g., differentiates contamination, colonization and clinical infection). Multiple samples also increase the yield and potential for culture growth. Mortality increases 8% with every 1 hour delay in the administration of appropriate antimicrobial therapy (ie. covers the actual organism). The initial pre antibiotic blood culture is often the only one that shows the causative organism. TIP Cultures: A positive tip culture with > 15 CFUs of an organism is used to identify that the catheter is the likely source of the positive culture (indicates high burden of organism attached to the catheter tip). |
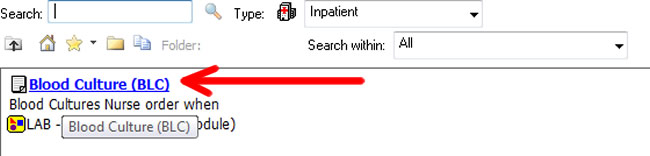
3. | Order Blood Cultures in Power Chart
Notes: Power Chart will not allow 2 identical lab tests to be ordered with the same time. |
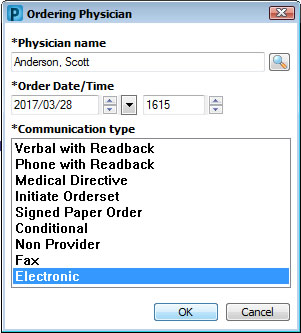
Enter ordering physician. Choose "electronic order" for Communication Type if you received a "nurse to order blood culture" communication order. Choose "electronic order" if you are reordering a previously entered order to provide more details Choose Medical Directive if you are initiating an order based on the Medical Directive guidelines | |
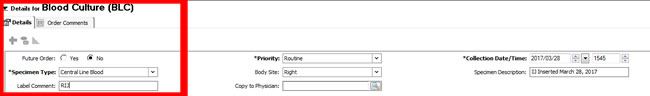
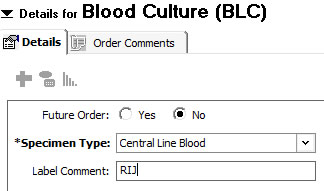
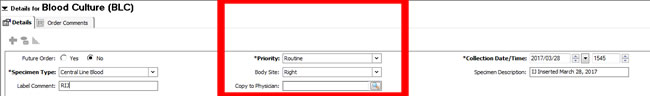
4. | Enter Specimen Type Under Specimen Type choose "Venipuncture", "Arterial Line Blood " or "Central Line Blood " from the drop down box Choose "Central Line Blood for a blood culture from a PICC, introducer, dialysis catheter or tunneled CVC In the Label Comment, type in the information that you would like to appear on your label that will help you to ensure that the label matches the sample Notes: Knowledge of the source of the culture is very important to the interpretation and treatment decision. |
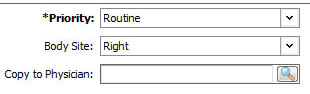
5. | Enter Priority and Body Site: In the Priority box, leave the default entry of "Routine". Blood cultures cannot be done STAT. From the BODY SITE drop down box, choose Left or Right (for speed of entry, type Left or Right) Notes: Interpretation of results and treatment decisions depends upon a clear understanding of the sample. Blood cultures require a minimum period of time for growth. Other specimens such as bronchoscopy sample or CSF specimen can be done STAT (this will provide a gram stain report). |
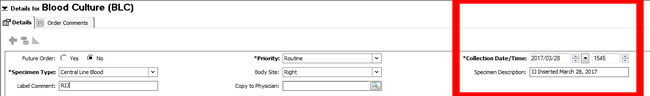
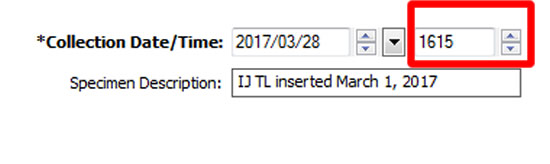
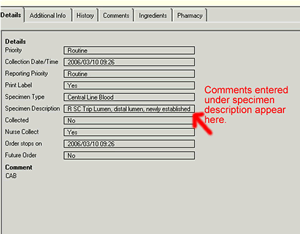
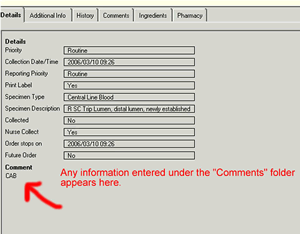
6. | Enter Collection Time and Specimen Description Adjust the ordering time for each blood culture sample (e.g., peripheral, arterial, central) to ensure they are at least one minute apart. In the Specimen Description box, enter the following information:
If this is a newly inserted line and the first sample being drawn, identify this as "newly inserted". Samples drawn at the time of line insertion may be labelled as "venipuncture" if needed for the purpose of obtaining a CAB assessment. |
| |
7. | Request Catheter Associated Bacteremia (CAB) Assessment If the patient has indwelling lines and the lines are not being immediately removed:
If the indwelling line is being removed, send cultures and a tip culture, but DO NOT request CAB assessment. CAB (Catheter Associated Bacteremia) assessment provides a "time to positivity result". If an indwelling line becomes positive by > 2 hours earlier than the peripheral sample, it suggests increased colonization of the catheter. If both the peripheral and line cultures become positive within 2 hours, it suggests bacteremia. Click to view Catheter Associated Bacteremia algorithm (only available from within LHSC). CAB assessment is not needed if the catheter is being removed. A tip culture provides a quantitative evaluation of organisms. A colony count > 15 plus a positive blood culture is indicative of Catheter Associated Bacteremia. |
8. | Collect Specimens Perform hand hygiene and don non-sterile gloves. Change the needleless access port prior to obtaining blood cultures Scrub the top of each specimen bottle with a separate swab Scrub vigorously in a horizontal direction using the first swab. Scrub vigorously in a vertical direction with the second swab. Allow the prep to dry FOR ONE FULL MINUTE before sampling. DO NOT DRAW A DISCARD SAMPLE UNLESS THERE IS CITRATE BLOCKING SOLUTION IN THE LINE. Collect a 10 ml sample for EACH bottle (discard volume is included in 10 ml sample). Citrate has antimicrobial properties, therefore, it should be removed from the sample before introducing it into the broth. Ensure that air does not enter the anaerobic bottle during collection. Remove non-sterile gloves and perform hand hygiene. Send samples down to lab in a biohazardous bag. Notes: Citrate may have antiseptic properties; a discard sample is required (e.g., for hemodialysis catheters). The back and forth scrubbing loosens bacteria and provides more effective disinfection of the site. The prep must dry to activate the antimicrobial properties. The Centre for Disease Control (CDC) recommends cleaning injection sites with 70% alcohol or iodophors. Chlorhexidine is recommended for skin preps and has not been studied for injection sites. Use of a product that includes alcohol is recommended. |
| 9. | Label Specimens Place labels on specimens. Select one of the small square barcodes and place on each bottle. Verify that the label name and patient are correct. Verify that the label matches the correct sample (e.g., arterial samples and arterial labels). Sign sample requisition and record time sample was drawn. Notes: Samples will be discarded by lab if unsigned. |
| 10. | Document Medical orders are required for blood sampling. Many routine lab tests can be ordered by a Critical Care Nurse by authority of Medical Directive. The reason for ordering a test or intervention by medical directive must be documented. |
| 11. | View Ordering Information Select the desired blood culture order from the from the Power Chart Flowsheet, Orders List or Task List. Right click and select "Orders Information" All of the information entered when the test was ordered is revealed. The name of the nurse who ordered the test is also displayed. Notes: Ordering information is available to aid in the interpretation of results.
|
| |
| Always log off, by exiting the patient's chart. | |
References: Blot, F., et al. (1999). Differential time to positivity between paired hub and peripheral blood culture (DTP) method. Lancet, 354: pp. 1071-1076. LHSC Policy for blood cultures Surviving Sepsis Guidelines 2016. Society of Critical Care Medicine. Developed by: Brenda Morgan, CNS, CCTC; Last Reviewed: January 17, 2025 |